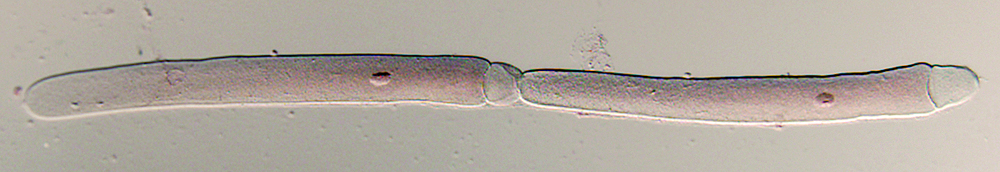
Specimen preparation and fixation
Why the coverslip method?
The easiest and most effective way to process gregarines for permanent mounts is by afixing them to coverslips and then processing the coverslips. We use 22x22mm coverslips because they are easy to handle and process. Some specialized tools are available. We purchase screw-cap Columbia coverslip staining jars for storing prepared smears and ceramic racks for coverslip staining. We make coverslip boxes to store cleaned coverslips for use.
Cleaning coverslips
The method depends on using a chemical fixative to coagulate the gregarine and gut fluid proteins in the smear so that they stick to the coverslip. This only works if the coverslips are clean. Despite what the vendor says, no coverslip is actually clean right out of the box. Thus it all begins with making clean coverslips. We clean coverslips individually by dipping them in 95% ethanol, polishing them with a dry cloth (we use bandanas), and blowing off any residual lint with compressed air. Rather than clean coverslips as they are needed, we store clean coverslips in lab-made boxes that keep out dust and debris. The plastic boxes are common, the plastic strips come in pairs as a free bonus in every box of Corning 2947 glass microslides. They're just trimmed and glued with cyanocrylate glue.
Fixatives
The object of chemical fixation is to kill the gregarines and permanently preserve their structure while introducing as little distortion or artifact as possible. There are many suitable fixatives, but we use Alchohol-Formalin-Acetic Acid (AFA) because it is fast, accurate, predictable, and simplifies the subsequent staining process. AFA works best for gregarines in the coverslip method when the fixative is hot, but not boiling. A standard 100-mm glass petri dish fits well on a coffe mug warming hot plate and keeps the AFA at about the right temperature. Cover the dish when not dropping or retrieving coverslips to reduce alcohol evaporation.
Dissection
There's nothing special about the microdissection technique for gregarines (#5 biology series forceps and a fine pair of scissors are recommended). As important is the use of a good physiological saline. Our standby saline is Belton & Grundfest saline, which is designed for beetles, but works pretty well with most insects. The saline is hypertonic for some insects, particularly odonates, and should be diluted 1:1 with water for use. Strategic use of tonicity is the handiest wrench in the microdissection toolbox. For example, adding distilled water to an odonate gut after it has already been removed and dissected in saline disrupts the insect epithelial cells much more quickly than the gregarines, freeing the complex gregarine epimerite from the intestinal epithelium. This allows you to make smears without breaking epimerites or having them occluded by host tissue. Practice and experimentation are the best teachers.
Making and fixing smears
We prepare a THIN smear of host intestine/gut fluid with gregarines on a clean coverslip and drop them face down into a petri-dish of fixative. The slips should float on the surface with no air bubbles. The surface tension of the fixative holds the gregarines against the glass until they are chemically affixed by protein coagulation. We let the slips float for at least 2 minutes and up to 15 minutes. Fixed specimen slips are individually removed with coverslip forceps and placed in a columbia jar of 70% ethanol for storage. Prepared slips can be stored in ethanol for months without seriously affecting subsequent staining.